AR Lab Network
Mountain Region
About us
The Antimicrobial Resistance Laboratory Network (AR Lab Network) at the Utah Public Health Laboratory (UPHL) was established by the Centers for Disease Control (CDC) in 2016. We serve as the Mountain States Regional Laboratory within the national AR Lab Network and receive specimens from Arizona, Colorado, Idaho, Montana , New Mexico, Utah, Texas, and Wyoming.
We screen, identify, and test bacterial and fungal samples from healthcare facilities and clinical laboratories to classify the kind of carbapenemase or antifungal resistance and prevent the spread of multidrug resistant organisms.
What we do
Identification
We confirm the identity of antimicrobial resistant isolates with the following methods:
- Bacterial identification using MALDI-TOF mass spectrometry
- Yeast (Candida spp) identification MALDI-TOF mass spectrometry
- Whole Genome Sequencing of isolates with suspected novel or emerging carbapenemase and to understand transmission.
Screening
We screen the following samples using culture and PCR methods for the presence of specific carbapenemases:
- CPOs such as CRE, CRPA, CRAB
- Candida auris
- Group A Streptococcus(Utah providers only)
We offer this service for free in collaboration with public health officials, such as state healthcare-associated Infections (HAI) coordinator, infection preventionists, and epidemiologists.
Antimicrobial Susceptibility (AST)
We perform antimicrobial susceptibility testing (AST) to confirm each sample’s phenotype and determine if it’s a multidrug-resistant organism.
Expanded AST (ExAST)
With pre-authorization, we perform expanded AST (ExAST) on highly resistant CRE, CRPA, and CRAB isolates that are Multidrug-Resistant Organism (MDROs).
Aztreonam-avibactam
Submit CRE Isolates that Test
- Not-susceptible to one carbapenem such as: Ertapenem, Meropenem, Doripenem, Imipenem, Ceftazidime-avibactam, Meropenem-vaborbactam, Imipenem- relebactam
- Or, test positive for NDM, VIM, or IMP genes
Meropenem-vaborbactam
Submit CRE Isolates that Test
- Not-susceptible to Meropenem
Imipenem-relebactam
Submit CRE (except for P. mirabilis) and CRPA that Test
- Not-susceptible to Imipenem
Sulbactam-durlobactam
Submit CRAB Isolates that Test
- Resistant to Imipenem, Meropenem or Doripenem
How to submit isolate or clinical (colonization screening) samples for testing
All samples, clinical and isolate, must be submitted to us with a completed test request form. Clinical samples must be collected with an ARLN-specific testing kit.
Follow these steps before submitting a sample to us:
- Contact us in advance to schedule testing and order testing kits.
- Email [email protected] if you are a laboratory or healthcare facility in Utah.
- Email [email protected] if you are a laboratory or healthcare facility in another Mountain Region state.
- Register with the Utah Lab Web Portal, please contact [email protected]
- Alternatively, fill out the ARLN Test Request Form
- Collect the samples and follow the instructions in the packaging and shipping guidance document when shipping clinical samples.
For clinical samples, you can also use the Lab Web Portal to fill out the test request form. If you would like to use Lab Web Portal, please contact [email protected]
Watch this video for more information on how to use the Lab Web Portal or review the Lab Web Portal Quick guide.
Submission, collection, and testing guidance
Colonization Collection guidance
- Colonization prioritization guidance
- CRE and CRPA Colonization specimen collection guidance
- CRAB specimen Colonization specimen collection guidance
- Group A Strep (GAS) Colonization Screening
- Candida auris Colonization specimen collection guidance
- General Information: CDC’s Candida auris Colonization screening
Isolate CRE, CRPA, and CRAB guidance
- Isolate: slant or plate
- CRE resistant to one or more carbapenem (ertapenem, doripenem, meropenem, or imipenem)
- CRPA resistant to a carbapenem (imipenem, meropenem) and non-susceptable to cefepime, ceftazidime, or ceftolozane-tazobactam
- CRAB resistant to any carbapenem (doripenem, imipenem, meropenem)
AST testing
Testing guidance:
Other resources:
ExAST testing
Testing guidelines
- Pre-authorization is required before submitting a sample for testing.
- We do not test multiple samples from the same patient, source, and organism in a 6-month period.
Pre-authorization
- AR Expanded AST Authorization Request Form.
- Email [email protected] your completed Expanded AST Authorization Request Form, and if you have questions about ExAST testing.
Candida spp. Isolate Submission and Updates, not albicans
- Isolate: slant or plate
- Candida isolate submission guidance updates
- CDC’s Send Candida Isolates
Neisseria gonorrhoeae
Testing guidelines
- Pre-authorization is required before submitting a sample for testing.
- We use E-test gradient strips (azithromycin, cefixime, ceftriaxone, and ciprofloxacin).
- Testing is for patients with suspected treatment failure or disseminated gonorrhea infections.
- We only accept bacterial isolates for testing.
Submission guidelines
- Resistant gonorrhea management document
- Submission guidelines for Neisseria gonorrhoeae gradient strip AST
Pre-authorization
- Email a completed test request form to [email protected].
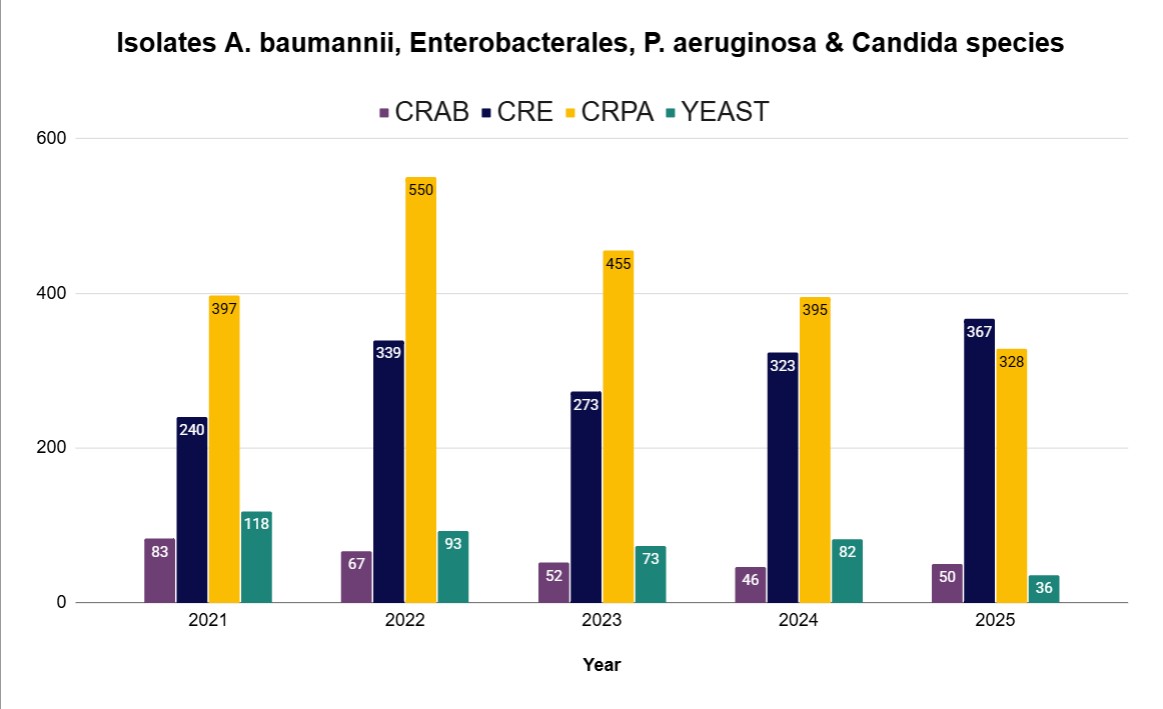
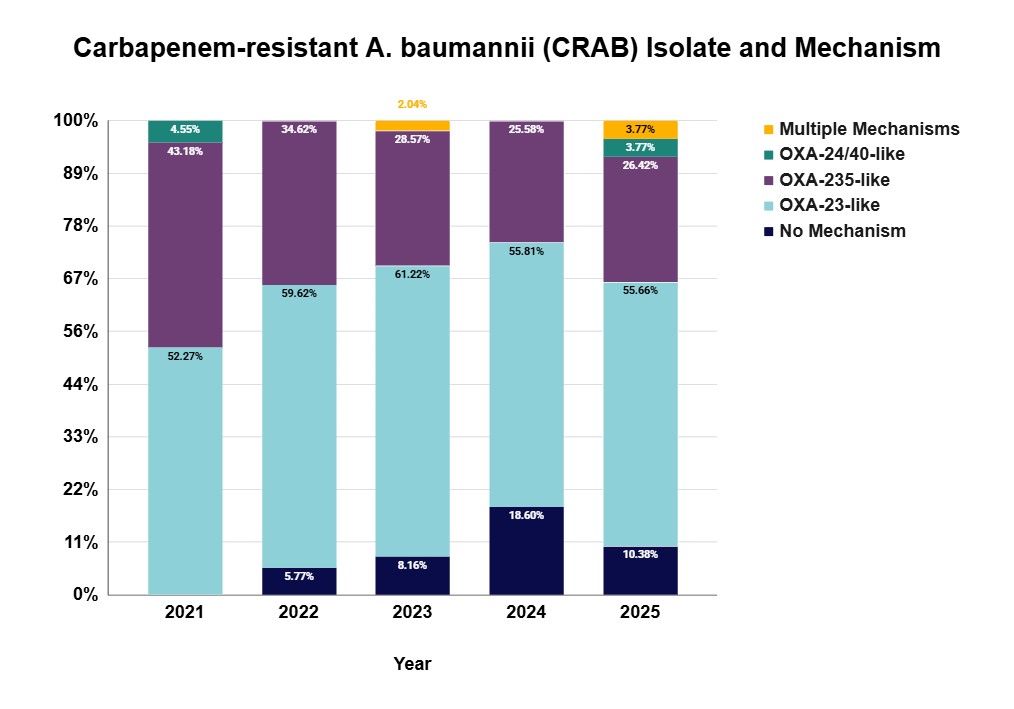
Data and reports
- AR lab, Mountain Region Report 2021-2023 (Q1 &Q2)
- AR lab, Mountain Region Report 2021-2023 (Q3 &Q4)
- Read the AR Lab, Mountain Region Report 20-21
- Mountain Region ARLN Newsletter 11/21
- AR Lab, Utah Situational Awareness 2024
- AR Lab, Utah Situational Awareness 2025 (Q1-Q4)
Contact us
For AR Lab Network questions
- AR Lab Network Mountain Region Laboratory; [email protected]
- Annette Atkinson, AR Lab Network Mountain Region Laboratory Manager; [email protected]
- Jesse Harbour, AR coordinator; [email protected]